Nagy, Brigitta and Szabados-Nacsa, Ágnes and Fülöp, Gergő and Nagyné Turák, Anikó and Galata, Dorián László and Farkas, Attila and Mészáros, Lilla Alexandra and Nagy, Zsombor Kristóf and Marosi, György (2023) Interpretable artificial neural networks for retrospective QbD of pharmaceutical tablet manufacturing based on a pilot-scale developmental dataset. INTERNATIONAL JOURNAL OF PHARMACEUTICS, 633. No.-122620. ISSN 0378-5173
|
Text
Cikk1_Interpretable.pdf - Published Version Available under License Creative Commons Attribution Non-commercial No Derivatives. Download (2MB) | Preview |
|
![[img]](http://real.mtak.hu/174554/7.hassmallThumbnailVersion/1-s2.0-S0378517323000406-ga1_lrg.jpg)
|
Text (Graphical abstract (high resolution))
1-s2.0-S0378517323000406-ga1_lrg.jpg Download (159kB) | Preview |
|
![[img]](http://real.mtak.hu/174554/8.hassmallThumbnailVersion/1-s2.0-S0378517323000406-ga1.jpg)
|
Text (Graphical abstract (full size))
1-s2.0-S0378517323000406-ga1.jpg Download (22kB) | Preview |
Abstract
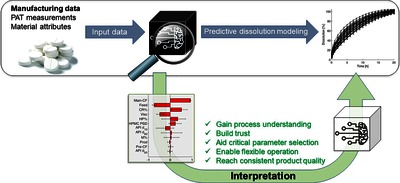
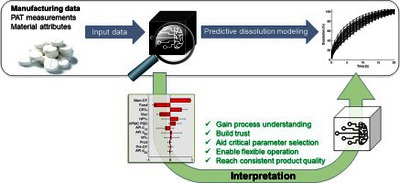
As the pharmaceutical industry increasingly adopts the Pharma 4.0. concept, there is a growing need to effectively predict the product quality based on manufacturing or in-process data. Although artificial neural networks (ANNs) have emerged as powerful tools in data-rich environments, their implementation in pharmaceutical manufacturing is hindered by their black-box nature. In this work, ANNs were developed and interpreted to demonstrate their applicability to increase process understanding by retrospective analysis of developmental or manufacturing data. The in vitro dissolution and hardness of extended-release, directly compressed tablets were predicted from manufacturing and spectroscopic data of pilot-scale development. The ANNs using material attributes and operational parameters provided better results than using NIR or Raman spectra as predictors. ANNs were interpreted by sensitivity analysis, helping to identify the root cause of the batch-to-batch variability, e.g., the variability in particle size, grade, or substitution of the hydroxypropyl methylcellulose excipient. An ANNbased control strategy was also successfully utilized to mitigate the batch-to-batch variability by flexibly operating the tableting process. The presented methodology can be adapted to arbitrary data-rich manufacturing steps from active substance synthesis to formulation to predict the quality from manufacturing or development data and gain process understanding and consistent product quality.
| Item Type: | Article |
|---|---|
| Uncontrolled Keywords: | Interpretable artificial neural networkExplainable artificial intelligenceRetrospective quality by designPharma 4.0Real-time release testingDissolution prediction |
| Subjects: | R Medicine / orvostudomány > RS Pharmacy and materia medica / gyógyszerészet, gyógyászati eszközök |
| SWORD Depositor: | MTMT SWORD |
| Depositing User: | MTMT SWORD |
| Date Deposited: | 23 Sep 2023 07:32 |
| Last Modified: | 23 Sep 2023 07:52 |
| URI: | http://real.mtak.hu/id/eprint/174554 |
Actions (login required)
 |
Edit Item |